Join Our Community of Healthcare Professionals
Training is provided online, accessible at any time, in easy to digest bitesize sessions, with a certificate awarded upon successful completion of a course. Our modules are aimed at both educating either new starters or those looking for refresher training on Department practices and procedures.
Department Subscriptions are available for the whole Team, with courses covering specific infection control and decontamination processes. For the Department Manager an overview of Team training is also provided through the Manager Dashboard.
Certified by the CPD Certification Service
Hospital iLearn courses are certified by the CPD Certification Service, pioneering CPD quality standards since 1996, and used by
NHS England.
Individual Subscriptions are also available for those looking to further their education on infection control and decontamination within a healthcare environment.
Annual Subscriptions
Courses & Features Providing Your Online Training Resource

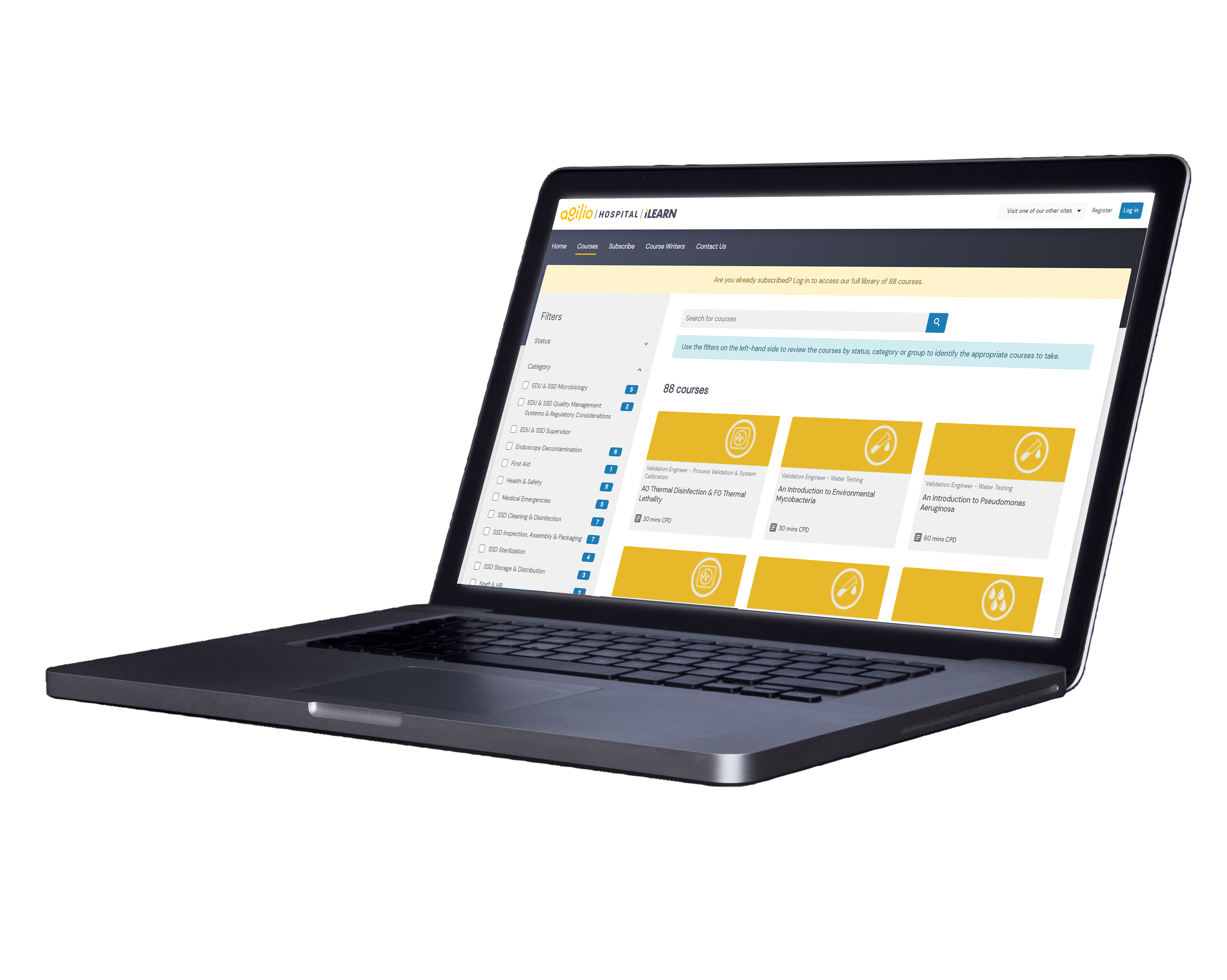
With over twelve specialist categories spanning a wide range of topics, staff have access to all training courses.

Each staff account allows storage of training certificates, gained either through Hospital iLearn or uploading external certificates, keeping everything in one place.

New courses are added monthly within our growing list of categories, with all courses reviewed and updated at least every 12 months, or whenever there is a change in standards and guidance.

Throughout your Department Subscription, we will be there to support you. We will automatically subscribe your Team, create and set up your Department and will be on hand for questions and support online or over the telephone.

As a Department Manager, your online dashboard provides a staff training register, along with course progression for each of your Team.

Download our Policy Documentation to Easily Incorporate Hospital iLearn into Your Department Training Procedures and QMS.
Membership & Pricing
Simple, straight-forward pricing. Choose the plan that's right for you.
- Access to ALL courses
- Department Manager Dashboard
- Access to ALL courses
- Certificate storage area
We offer courses for all your Department needs
Regularly updated and written by experts we pride ourselves in providing quality training guiding you to compliance
Some of our courses
This course explains the different stages for endoscopy decontamination including manual cleaning and using an EWD.
This course provides an understanding of the various wash processes completed in the wash area. Covering prewash, manual wash, and automated water disinfectors. Also, an understanding into water types and detergents.
This course explains what a Quality Management System (QMS) is, how it works and the importance of how it relates to your role.